The groups of Anthony Kossiakoff, Eduardo Perozo, and Shohei Koide at the University of Chicago determined the crystal structure of the full-length potassium channel, KcsA, in its closed conformation. The tetrameric, proton-activated, voltage-modulated K+ channel has provided the highest-resolution structures of ion channels to date. However, previous KcsA structures lacked a C-terminal domain (residues 125-160) that stabilizes the closed form of the channel. With the aid of synthetic Fabs, full-length KcsA was crystallized, revealing a 4-helix bundle extending about 70 Å into the cytoplasm. The bundle promotes a 15 degree bend in the inner bundle gate, tightening its diameter from about 4 Å to about 2.5 Å, and shifting the narrowest point by two helical turns into the membrane bilayer. The 4-helix bundle is proposed to remain intact during gating, and the structure to represent the physiologically relevant closed state of KcsA.
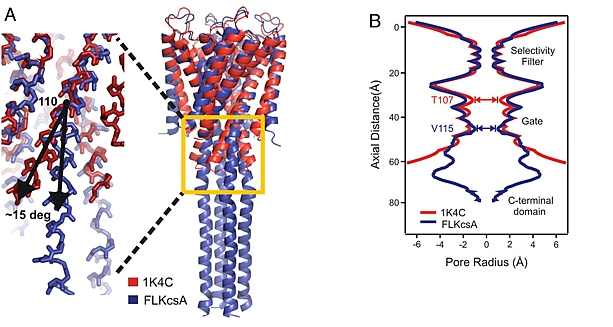 |
Figure: Influence of the C-terminal truncation on the conformation of the inner helix gate of KcsA. (A) Cα superposition of the high-resolution truncated KcsA structure (1K4C; red) with full-length KcsA (blue). Inset highlights the splaying out of the inner helix bundle gate between residues 110 and 115, resulting in a 15 degree outward tilting. (B) Radius profile of truncated (red) and full-length KcsA (blue). |
Citation:
Uysal, S, Vasquez, V, Tereshko, V, Esaki, K, Fellouse, FA, Sidhu, SS, Koide,
S, Perozo, E, Kossiakoff, A., Crystal structure of full-length KcsA in its
closed conformation, Proc. Natl. Acad. Sci. USA 106 (16), 6644-6649 (2009).
DOI: 10.1073/pnas.0810663106