Ian Wilson's group at the Scripps Research Institute solved structures of a broadly neutralizing antibody complexed to hemagglutinin (HA) from two different strains of influenza virus. HA allows the flu virus to bind target cells, but its structure changes from strain to strain, which presents a great challenge for vaccine development. The protein contains two principal regions: a head that varies among strains and a more conserved stalk. Many antibodies bind to the HA head, but these are effective against only a limited range of viral strains. Current configurations of the flu vaccine inoculate recipients against three of the major influenza subtypes: the virus that caused the infamous Spanish flu of 1918 and the viruses that caused the flu pandemics of 1957 and 1968. The study showed that a recently isolated, broadly neutralizing antibody binds to a conserved helical region of the stalk of HAs from two different strains of the virus - that of the 1913 H1N1 pandemic, and that of a recent lethal case of H5N1 avian flu. These results may allow vaccine development targeted to the conserved stalk regions of HA.
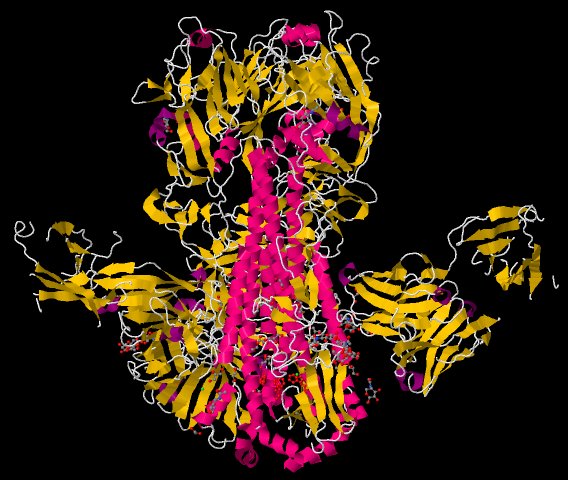 |
Figure: Structure of Fab CR6261 in Complex with the 1918 H1N1 influenza virus hemagglutinin [PDB ID 3GBN] |
Citation:
Ekiert, DC, Bhabha, G, Elsliger, M-A, Friesen, RHE, Jongeneelen, M, Throsby,
M, Goudsmit, J, Wilson, IA. Antibody Recognition of a Highly Conserved
Influenza Virus Epitope, Science 324, 246-251 (2009). DOI:
10.1126/science.1171491