The group of Ian Wilson at The Scripps Research Institute produced important structural work on the immune response to bacterial infection. The vertebrate immune system senses invasion of diverse types of flagellated bacteria using Toll-like receptor 5 (TLR5). TLR5 interacts with a flagellin protein that constitutes bacterial flagella, subsequently triggering the first line of defense to clear flagellated pathogens from our bodies. It is notoriously difficult to work with TLR5, but the Wilson group managed to solve the 2.47-Å crystal structure of a complex between the N-terminal fragment of zebrafish TLR5 and the Salmonella flagellin D1-D2-D3 domains. TLR5 forms a 2:2 complex with flagellin via homodimerization of two copies of 1:1 TLR5-flagellin heterodimers. TLR5 makes specific polar contacts with highly conserved residues in the D1 domain of flagellin, which provides an explanation for activation of TLR5 signaling by a variety of flagellins. The assembly of two TLR5 chains in the complex in a tail-to-tail orientation resembles other TLRs that bind non-protein ligands, in which the proximal C-terminal regions in the middle of the complex initiate signaling. Structural information on the TLR5-flagellin interaction at atomic resolution provides valuable insights into development of novel vaccine adjuvants and advancement of therapeutic applications for hyper-inflammatory diseases.
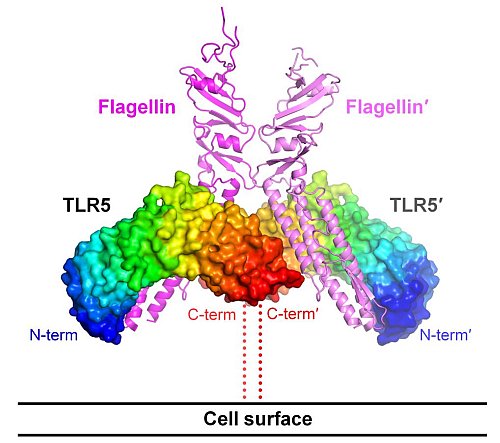 |
Figure: Crystal structure of the 2:2 TLR5-flagellin complex. Two basic units of 1:1 heterodimers (TLR5-flagellin and TLR5'-flagellin') assemble into a 2:2 complex, organizing two TLR5 chains in a tail-to-tail orientation. |
Citation:
Yoon, SI, Kurnasov, O, Natarajan, V, Hong, M, Gudkov, AV, Osterman, AL,
Wilson, IA. Structural basis of TLR5-flagellin recognition and signaling,
Science 335, 859-864 (2012).