Jue Chen's group at Purdue University determined the structure of the multidrug transporter P-glycoprotein with accuracy sufficient to reveal drug binding sites. P-glycoprotein (P-gp) is a multi-drug transporter responsible for most clinical resistance to chemotherapeutic agents in cancer treatment. In addition, P-gp also affects the absorption, distribution, and clearance of many clinically important drugs unrelated to cancer. In the pharmaceutical industry it is common practice to evaluate at an early stage of development a compound's susceptibility to clearance by P-gp. The researchers determined the crystal structure of P-gp from C. elegans to 3.4-Å resolution and established its accuracy with 34 selenium-labeled methionines and 3 mercury-labeled cysteines. The protein is 46% identical to the human ortholog and it exhibits many of the same functional properties, including cellular resistance to the cancer chemotherapeutic agents taxol and actinomycin D. The structure lends itself immediately to an understanding of drug access from the membrane to the transport pathway and to a mechanistic view of coupling between the nucleotide-binding domains and the drug transport pathway.
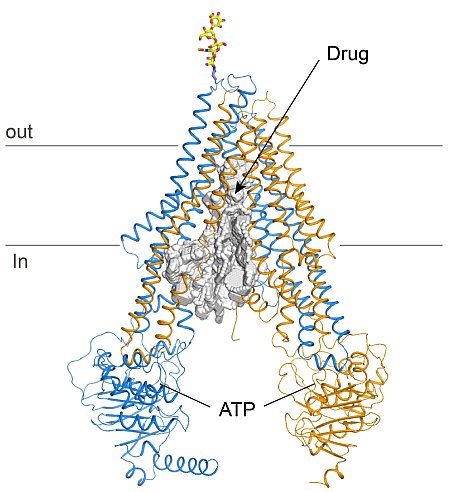 |
Figure: The crystal structure of C. elegans P-glycoprotein. The N- and C- terminal halves of P-glycoprotein are shown in cyan and orange, respectively. An oligosaccharide attached at N125 is shown in stick model. The binding sites for ATP and drug are indicated. |
Citation:
Jin, MS, Oldham, ML, Zhang, Q, Chen, J. Crystal structure of the multidrug
transporter P-glycoprotein from Caenorhabditis elegans, Nature 490, 566-569
(2012).