 |
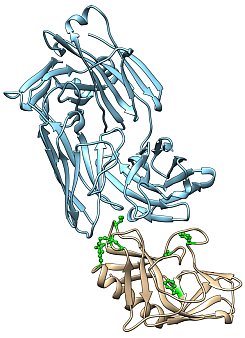
Figure: Engineered 60-mer protein-based nanoparticle immunogen, based on broadly neutralizing anti-HIV antibodies. |
GM/CA@APS contributed in part to ground-breaking research on HIV vaccine development that was recently featured on the cover of Science, (Jardine, Julien, Menis et al., 2013). Inducing broadly neutralizing antibodies (bNAbs) against HIV-1 through vaccination is a major goal for ongoing vaccine development efforts and constitutes a global health priority. Several bNAbs have been isolated from seropositive individuals but these bNAbs often take more than two years to develop. However, these bNAbs are able to protect from HIV-1 acquisition in animal models when they are given prior to virus exposure. This provides proof-of-principle that, if these bNAbs could be re-elicited by vaccination, they should protect against HIV-1 acquisition. One class of these bNAbs, named the VRC01-class, targets the CD4-receptor binding site on the viral glycoprotein Env, which is responsible for HIV-1 entry into host cells. A major hurdle in re-eliciting VRC01-class bNAbs by vaccination resides in the inability of naturally circulating Env to readily activate VRC01-class germline B-cell precursors. To reliably target VRC01-class germline B-cell precursors by vaccination and initiate a productive immune response, the Schief lab at The Scripps Research Institute used computation-guided, in vitro screening to engineer a germline-targeting gp120 outer domain immunogen that binds to germline precursors, as well as to multiple VRC01-class bNAbs. Using GM/CA@APS beamlines, the lab of Ian Wilson elucidated various crystal structures that explained the inability of VRC01-class antibody precursors to interact with Env. This structural information was essential in guiding design of improved antigens, as well as in assisting in engineering a 60-mer protein-based nanoparticle that readily activates germline and mature VRC01-class B cells in vitro. These 60-mer nanoparticle immunogens are currently under study for their ability to kick-start a productive immune response in various animal models. In principle, this structure-guided immunogen design strategy to specifically target germline precursors of bNAbs could also be applied to other epitopes and pathogens.
Citation:
Jardine, J, Julien, JP, Menis, S, Ota, T, Kalyuzhniy, O, McGuire, A, Sok, D,
Huang, PS, MacPherson, S, Jones, M, Nieusma, T, Mathison, J, Baker, D, Ward,
AB, Burton, DR, Stamatatos, L, Nemazee, D, Wilson, IA, Schief, WR. Rational
HIV immunogen design to target specific germline B cell receptors, Science
340, 711-716 (2013).