The group of Jochen Zimmer at the University of Virginia determined the structure of a translocation intermediate of a membrane protein involved in cellulose synthesis and transport. Cellulose, the most abundant biological macromolecule on the planet, is an extracellular, linear polymer of glucose molecules. It represents an essential component of plant cell walls but is also found in algae and bacteria. In bacteria, cellulose production frequently correlates with the formation of biofilms, a sessile, multicellular growth form. Cellulose synthesis and transport across the inner bacterial membrane is mediated by a complex of the multi-spanning catalytic BcsA subunit and the membrane-anchored, periplasmic BcsB protein. The authors determined the crystal structure of a remarkable complex of BcsA and BcsB from Rhodobacter sphaeroides containing a translocating polysaccharide. The structure of the BcsA-B translocation intermediate reveals the architecture of the cellulose synthase, demonstrates how BcsA forms a cellulose-conducting channel, and suggests a model for the coupling of cellulose synthesis and translocation in which the nascent polysaccharide is extended by one glucose molecule at a time.
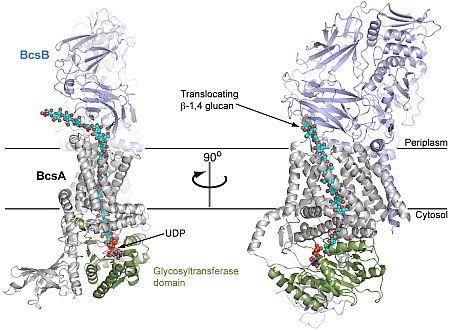 |
Figure: Orthogonal views of BcsA-B translocation intermediate, showing a translocating polysaccharide. |
Citation: Morgan JLW, Strumillo J, Zimmer J. Crystallographic snapshot of cellulose synthesis and membrane translocation. Nature. 2013 January 10; 493: 181-186. doi:10.1038/nature11744