The group of Daniel Rosenbaum at University of Texas Southwestern Medical Center determined the structure of the human subtype 2 orexin receptor (OX2R), a physiologically important GPCR in the central nervous system. The human OX2R mediates the activity of the orexin neurotransmitters, which are critical modulators of sleep, arousal and other behaviors in mammals. The OX2R structure was solved in the presence of the drug suvorexant, the first-in-class breakthrough orexin receptor antagonist recently approved by the FDA for treatment of insomnia. Defects in orexin signaling are responsible for the human disease narcolepsy, and there are currently no drugs that act as orexin receptor agonists. This paper represented the first structure of an orexin receptor, which was solved by a combination of protein engineering, lipid-mediated crystallization, and use of GM/CA@APS beamlines. The structure reveals how suvorexant adopts a compact folded conformation and binds to the receptor deep in the orthosteric pocket, stabilizing a network of extracellular salt bridges and blocking transmembrane helix motions necessary for activation. Elucidation of the molecular architecture of the human OX2R expands our understanding of peptidergic GPCR ligand recognition and will aid further efforts to modulate orexin signaling for therapeutic ends.
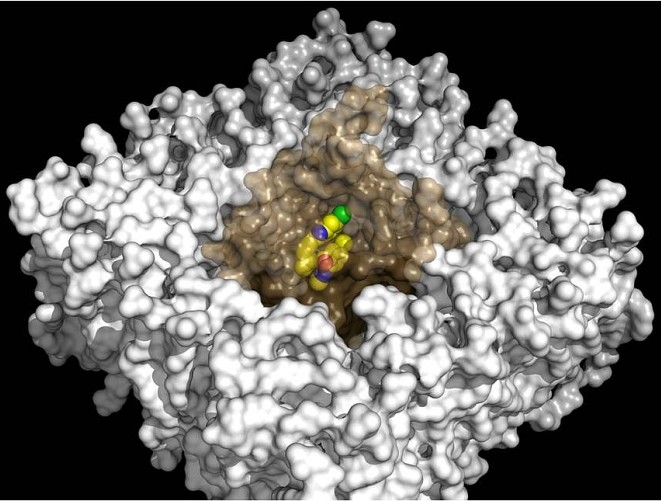 |
Figure: Surface rendering of the human OX2R orexin receptor (brown) bound to suvorexant (yellow spheres) embedded within a model phospholipid membrane (white). |
Citation: Jie Yin, Juan Carlos Mobarec, Peter Kolb, Daniel M. Rosenbaum , "Crystal structure of the human OX2 orexin receptor bound to the insomnia drug suvorexant," Nature 518, 247-250 (2014). DOI: 10.1038/nature14035