Type 2 diabetes mellitus (T2DM) affects hundreds of millions of people worldwide. The human receptor protein GPR40 has been identified by the Takeda pharmaceutical company as a potential new drug target. GPR40 is a fatty-acid receptor that enhances glucose-dependent insulin secretion upon ligand binding. TAK-875 (fasiglifam), a partial agonist of GPR40, was brought into clinical development by Takeda as a possible new treatment for T2DM. To better understand the mode of action of TAK-875, the three-dimensional structure of GPR40 bound to TAK-875 was determined Gyorgy Snell and colleagues at Takeda after overcoming a number of challenging experimental hurdles. The GPR40 crystals were small - about 20-30 microns - and were grown in a media that makes them barely visible to optical microscopes. Raster screening was used extensively in combination with the 20 micron mini-beam at GM/CA@APS. Using data collected on a dozen crystals at 23ID-B and 23ID-D, and a dataset collected on a single crystal at beamline 5.0.3 of the Advanced Light Source, the structure of the complex was determined at 2.3-Å resolution. The structure provided surprising insights into how the receptor and the drug interact. TAK-875 binds in a channel flanked by transmembrane helices 3 and 4, and not in the ligand binding site usually observed in other GPCR structures (called the orthosteric binding site). The structure is consistent with TAK-875 entering the receptor through the lipid bilayer and provides a snapshot of the extended charge network involved in receptor-drug interaction.
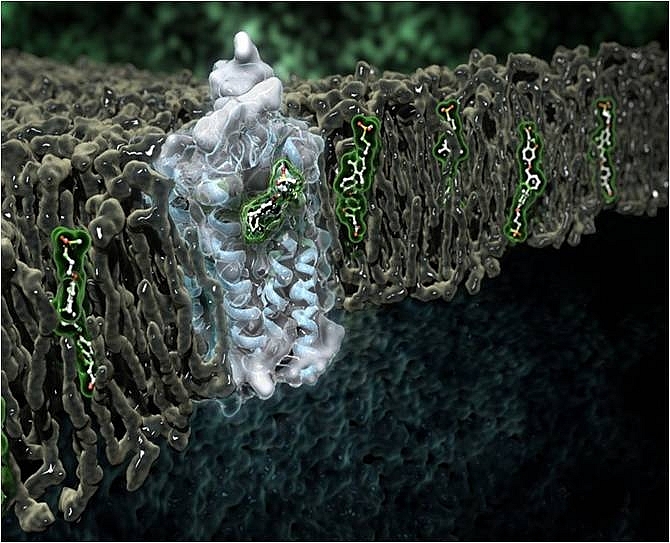 |
Figure: Artist's rendering of the GPR40-TAK-875 complex inside the cell membrane. |
Citation: Ankita Srivastava, Jason Yano, Yoshihiko Hirozane, Georgia Kefala, Franz Gruswitz, Gyorgy Snell, Weston Lane, Anthony Ivetac, Kathleen Aertgeerts, Jasmine Nguyen, Andy Jennings, Kengo Okada. "High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875," Nature 513, 124-127 (2014). DOI: 10.1038/nature13494