 |
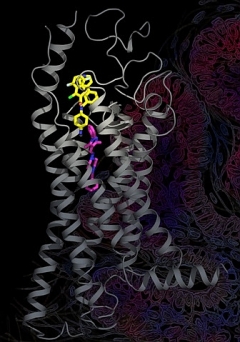
Figure: Structure of the smoothened receptor demonstrating its extended ligand binding site. The antagonist SANT1 (magenta) binds deeper into the pocket than do two other ligands (yellow), the antagonist Anta XV and the agonist SAG1.5. |
The Ray Stevens group from The Scripps Research Institute and collaborators from Duke, the NIMH, and the University of North Carolina determined structures of the smoothened receptor (SMO) bound to two antagonists and one agonist. SMO mediates signal transduction in the hedgehog (Hh) pathway, which plays an important role in normal embryonic development; aberrant SMO activation can promote carcinogenesis and it is therefore a target of anticancer drugs. SMO is a frizzled family (or class F) GPCR. The study generated new insights into the conformational plasticity of SMO-ligand interactions by crystallographic and biochemical characterization of several SMO complexes with diverse ligand chemotypes. Importantly, these ligands target distinct sites in the elongated cavity, have different interactions with known drug-resistant mutants and include both antagonists and agonists of Hh signalling. The details of these interactions and ligand-dependent conformational changes, correlated with specific functional features of the ligands, will help to design new clinical candidates targeting SMO with attenuated side effects and chemoresistance.
Citation: Wang C, Wu H, Evron T, Vardy E, Han GW, Huang XP, Hufeisen SJ, Mangano TJ, Urban DJ, Katritch V, Cherezov V, Caron MG, Roth BL, Stevens RC. Structural basis for Smoothened receptor modulation and chemoresistance to anticancer drugs. Nat Commun. 2014 Jul 10;5:4355. doi: 10.1038/ncomms5355.