 |
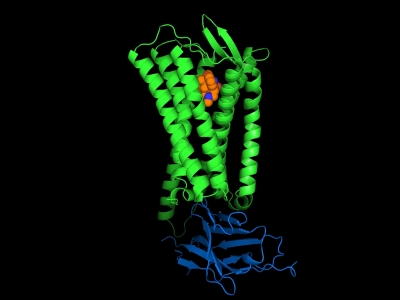
Figure: μ-opioid receptor (green) bound to the agonist BU72 (orange) and a G-protein mimetic antibody fragment (blue) |
The groups of Brian Kobilka, Bill Weis and Ron Dror (Stanford University) investigated the structural basis for μ-opioid receptor (μOR) activation. Opioid receptors in the central nervous system mediate responses to opioid alkaloids and related pharmaceuticals to produce a wide range of effects including analgesia, euphoria, sedation, respiratory depression and cough suppression, as well as gastrointestinal effects. Opioid receptors were classified into four subclasses including μ-OR, δ-OR, κ-OR and the nociceptin receptor. The μOR is the primary target for the majority of clinically approved opioid analgesics as well as drugs of abuse. To better understand the activation mechanism and to facilitate the development of analgesic drugs with fewer side effects, the group determined the structure of μ-opioid receptor bound to agonist BU72 and stabilized in the active state with a G-protein mimetic camelid antibody fragment (Nb39). Compared with the inactive state μOR, the 2.1 Å active-state μOR structure revealed how subtle changes in the ligand binding site are associated with large conformational changes in the cytoplasmic surface. The active μOR structure also revealed an extensive polar network, which may explain the inefficient allosteric coupling of the orthosteric pocket and the G-protein-coupling interface observed in other studies for both the μOR and the β2 adrenergic receptor. The μOR structure offers important insights into shared and subtype-specific mechanisms for the activation of family A GPCRs.
Citation: Huang W, Manglik A, Venkatakrishnan AJ, Laeremans T, Feinberg EN, Sanborn AL, Kato HE, Livingston KE, Thorsen TS, Kling R, Granier S, Gmeiner P, Husbands SM, Traynor JR, Weis WI, Steyaert J, Dror RO, Kobilka BK. (2015) Structural insights into μ-opioid receptor activation. Nature 524, 315-321.