Using data from beamline 23ID-D, the group of Pam Bjorkman (California Institute of Technology) determined the structure of the HIV broadly neutralizing antibody (bNab) 8ANC195 bound to an HIV envelope trimer. The 8ANC195 epitope, defined by crystallography with bNab 8ANC195 complexed with monomeric gp120, and by EM structures of bNAb 8ANC195 complexed with Env, spans the gp120 and gp41 Env subunits. To investigate 8ANC195's gp41 epitope at higher resolution, the researchers solved the crystal structure of 8ANC195 complexes with fully glycosylated Env trimer at 3.58 Å resolution, revealing that 8ANC195 inserts into a glycan shield gap and contacts glycans and amino acids on both gp120 and gp41. To determine whether 8ANC195 recognizes the CD4-bound open Env conformation that leads to co-receptor binding and fusion, one of several known conformations of virion-associated Env, they also solved EM structures of an Env/CD4/CD4-induced antibody/8ANC195 complex. 8ANC195 binding partially closed the CD4-bound trimer, which highlights the structural plasticity of Env and reveals a previously unseen conformation. The 3.58 Å X-ray crystal structure of the 8ANC195-Env trimer defines the gp120-gp41 site of HIV vulnerability. The ability of 8ANC195 to bind both closed and open Env conformations suggests advantages for potential therapeutic applications.
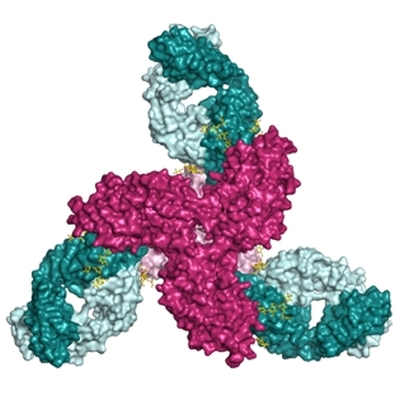 |
Figure: HIV envelope trimer bound to a broadly neutralizing antibody. 8ANC195 (aquamarine and light blue), gp120 trimer subunits (cranberry), gp41 subunits (pink). N-linked glycans at the Fab-trimer interface (yellow and red). |
Citation: Scharf, L., Wang, H., Gao, H., Chen, S., McDowall, A. W., and Bjorkman, P. J. (2015) Broadly neutralizing antibody 8ANC195 recognizes closed and open states of HIV-1 Env, Cell 162, 1379-1390.