The group of Sylvie Doublie at the University of Vermont and collaborators determined the first crystal structure of human DNA polymerase theta (pol theta) complexed to nucleotides and lesion-containing DNA. The human POLQ gene encodes pol theta, a specialized family-A DNA polymerase over-expressed in breast and ovarian cancers. Pol theta participates in an alternative end-joining pathway to repair DNA double-strand breaks, thus contributing to genome stability. This work uncovers the basis of pol theta specialization, demonstrating how conserved arginine and lysine amino acids of the thumb subdomain enhance the protein's hold on the DNA phosphate backbone to mediate end-joining. One such arginine (Arg2254) is uniquely poised to stabilize the primer terminus and is absolutely required for efficient bypass of non-templating DNA lesions. The structures also demonstrate potential for transient higher-order pol theta complexes, which could provide a novel route for pharmacological inhibition in future cancer therapies.
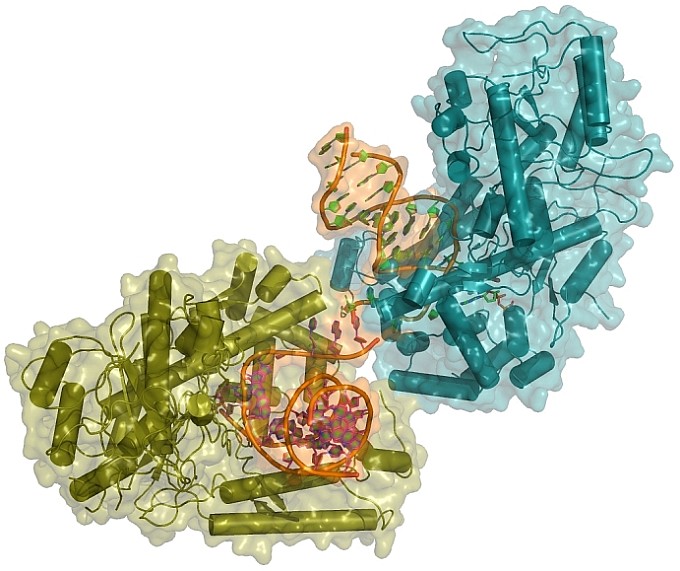 |
Figure: Two molecules of Human DNA pol theta bind lesion containing double-stranded DNA. |
Citation: Zahn KE, Averill AM, Aller P, Wood RD, Doublie S. Human DNA polymerase θ grasps the primer terminus to mediate DNA repair. Nat Struct Mol Biol. 2015 Apr;22(4):304-11.