In a study by Yi Zheng led by Tracy Handel and Irina Kufareva at the University of California San Diego and collaborators from the University of Leiden, Bristol Meyers Squibb and the University of Southern California, a structure of the CC chemokine receptor 2 (CCR2) was solved in complex with simultaneously bound orthosteric and allosteric antagonists. Chemokine receptors are GPCRs that regulate cell migration. CCR2 plays a role in numerous disease states including atherosclerosis, multiple sclerosis, asthma, neuropathic pain, diabetic nephropathy, and cancer metastasis.
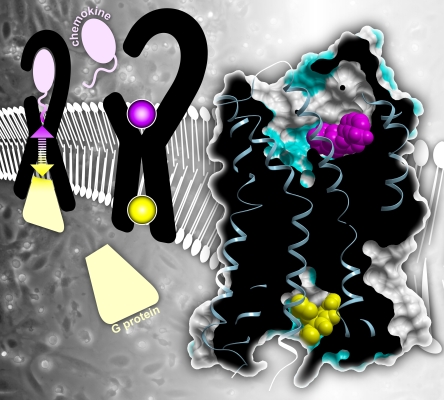 |
Schematic shows that upon binding of a chemokine (pink) to the extracellular side of the chemokine receptor (black), signals are transmitted to the intracellular side where G proteins (yellow) are activated. The communication is two-way (two-headed arrow) because G proteins also enhance receptor chemokine affinity. The middle image shows that chemokine receptors can be inhibited by compounds that bind to an orthosteric site on the extracellular side of the receptor to directly block the chemokine, or by those that bind to an allosteric site on the intracellular side to block the G protein directly and the chemokine indirectly. The right image shows the cooperative binding of orthosteric and allosteric antagonists, indicating that allosteric communication is relevant to receptor inhibition as well as activation. The allosteric antagonist binds in a novel pocket. Such allosteric inhibitors may prove to be particularly useful drugs because of their unique mechanisms of action. |
Citation: Zheng Y, Qin L, Ortiz Zacarias NV, de Vries H, Han GW, Gustavsson M, Dabros M, Zhao C, Cherney RJ, Carter P, Stamos D, Abagyan R, Cherezov V, Stevens RC, IJzerman AP, Heitman LH, Tebben A, Kufareva I, Handel TM, "Structure of CC chemokine receptor 2 with orthosteric and allosteric antagonists," Nature 540, 458-461 (2016). DOI: 10.1038/nature20605.