 |
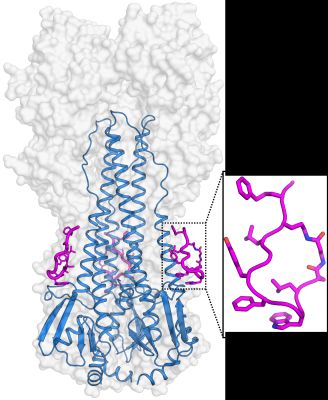
Figure: the crystal structure of a complex between a cyclic peptide (magenta) and influenza virus hemagglutinin (blue with grey surface) |
The group of Ian Wilson at The Scripps Research Institute, in collaboration with a team of researchers from Janssen Research & Development (Janssen, Netherlands), discovered and analyzed potent peptidic fusion inhibitors of influenza virus. The peptide designs were based on the broadly neutralizing antibodies (bnAbs) CR9114 and FI6v3, which target the highly conserved stem epitope on influenza virus hemagglutinin (HA). The designed peptides show high affinity and broad neutralization against group 1 influenza A viruses. High-resolution crystal structures of the peptides in complex with HA from A/Puerto Rico/8/1934 (H1N1, H1/PR8) were determined. These demonstrated that the peptides target a highly conserved hydrophobic pocket in the HA stem region, as designed, and recapitulate the binding mode of the bnAbs CR9114 and FI6v3 on the HA. The designed peptides stabilize the HA trimer and prevent the low-pH-induced conformational changes in the endosome that initiate a flu infection by fusion of viral and host cell membranes. This structural information on the peptide-HA complexes provides valuable insights for influenza virus drug development targeting hemagglutinin.
Citation: Kadam RU, Juraszek J, Brandenburg B, Buyck C, Schepens WBG, Kesteleyn B, Stoops B, Vreeken RJ, Vermond J, Goutier W, Tang C, Vogels R, Friesen RHE, Goudsmit J, van Dongen MJP, Wilson IA, Potent peptidic fusion inhibitors of influenza virus, Science 358, 496-502 (2017). DOI: 10.1126/science.aan0516