As a fundamental pathway in embryonic development and tissue homeostasis, hyperactivation of the Hedgehog signaling pathway can lead to cancers such as medulloblastoma (MB) and basal cell carcinoma (BCC). These cancers are frequently driven by activating mutations in Smoothened (SMO), a class F (Frizzled) GPCR superfamily receptor that acts as the key signal transducer in the pathway. In order to understand the molecular basis of SMO activation, the group of Adrian Salic, in collaboration with the group of Andrew Kruse, both at Harvard Medical School, determined two crystal structures of sterol-activated SMO including its endogenous agonist cholesterol, demonstrating for the first time, the active conformation of a class F GPCR. Cholesterol binds to and induces a dramatic reorientation of the extracellular cysteine-rich domain (CRD), which is then relayed to activate the 7-transmembrane domain (7TM) of SMO, causing its outward opening, in a manner reminiscent of classic GPCRs upon activation. In absence of stimulation, human SMO is autoinhibited by a unique pi-cation lock, which is composed of R451 from TM6 and W535 from TM7, keeping the 7TM in the "closed" inactive conformation to prevent undesired signaling. Cholesterol binding allosterically breaks the lock, resulting in the outward movement of TM6 to drive SMO activation. Interestingly, W535L is an oncogenic SMO-M2 mutant identified in sporadic BCC that disrupts such inhibitory interaction, thus providing an explanation for its constitutive activity. Unexpectedly, the structures also reveal a longitudinal hydrophobic tunnel in SMO, suggesting a cholesterol transport pathway from the inner membrane leaflet to the CRD, whereby it triggers SMO activation. These results help answer a long-standing mystery in Hedgehog signal transduction through SMO, and also provide a structural basis defining the mechanism for small-molecule SMO antagonism, which may promise therapeutic strategies to overcome clinical SMO inhibitor resistance in cancers.
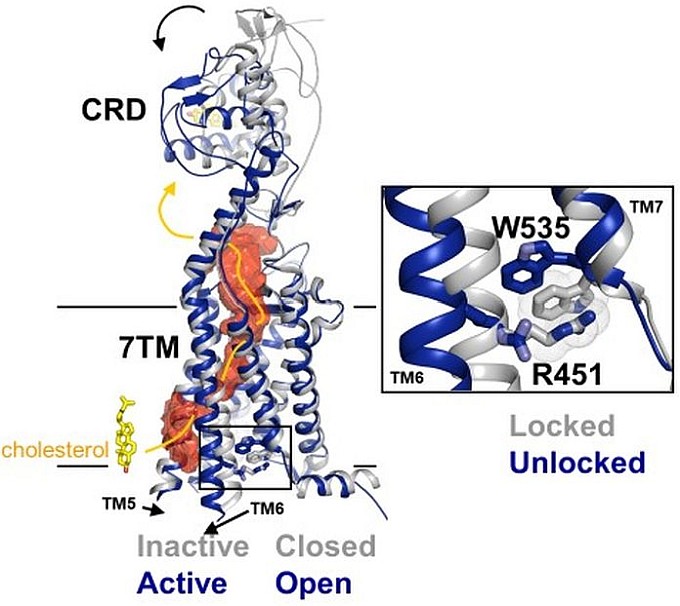 |
Figure: Cholesterol-triggered Smoothened activation in Hedgehog signaling (left); the pi-cation lock (right). |
Citation: Huang, P, Zheng, S, Wierbowski, BM, Kim, Y, Nedelcu, D, Aravena, L, Liu, J, Kruse, AC, Salic, A., Structural basis of Smoothened activation in Hedgehog signaling, Cell 174, 312-324 (2018). DOI: 10.1016/j.cell.2018.04.029