The group of Amie Boal at The Pennsylvania State University, in collaboration with Emily Balskus (Harvard University) and her research team, determined crystal structures of an enzyme, SznF, responsible for constructing an N-nitrosamine functional group in the anticancer drug, streptozotocin (SZN). While used in the clinic as a pancreatic cancer drug for more than 35 years, the biosynthesis of SZN remained unknown until recently. The Balskus group identified the genes for SZN production in a soil bacterium and showed that the pharmacologically active N-nitrosamine unit originates from a modified L-arginine amino acid precursor. The structure of SznF showed that the enzyme contains two active sites with different non-heme iron cofactors and reactivities. The N-nitrosamine unit is formed in the last step of the SznF reaction via an O2-dependent rearrangement with little precedent in biological or synthetic chemistry. A structure of SznF with an analog of the rearrangement substrate shows that the precursor compound binds directly to the iron cofactor responsible for the transformation. This coordination mode reveals a strategy for choreographing the complex bond-breaking and bond-forming steps that must occur to synthesize the key bioactive functional group of SZN. The study also shows that more than 300 SznF proteins exist in diverse bacteria, including important human pathogens and plant symbionts. This discovery suggests that bioactive N-nitroso compounds may have previously unrecognized roles in bacterial infection and mutualism.
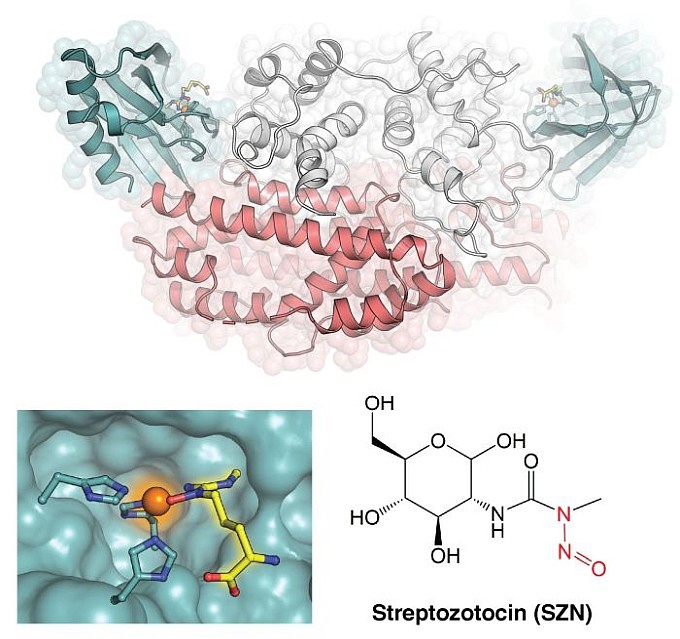 |
Figure: Homodimer structure of SznF (top); a substrate analog (yellow) bound to an iron cofactor (bottom left); the pancreatic cancer drug, SZN (lower right). |
Citation: Ng, TL, Rohac, R, Mitchell, AJ, Boal, AK, Balskus, EP. An N-nitrosating metalloenzyme constructs the pharmacophore of streptozotocin, Nature 566, 94-99 (2019). DOI: 10.1038/s41586-019-0894-z.