Matt Whorton's group of the Oregon Health & Science University determined X-ray crystal structures of a mammalian nucleotide-sugar transporter (NST) - the mouse CMP-sialic acid transporter (mCST) - in complex with its physiological ligands, cytidine monophosphate (CMP) and CMP-sialic acid (CMP-Sia). NSTs are primarily expressed in the Golgi membrane where they transport nucleotide-activated sugars from the cytoplasm to the lumen of the Golgi. Here, the sugars are used by glycosyltransferase enzymes to build glycan chains that are attached to proteins and lipids. Humans have seven types of NSTs which each specifically transport only one or two of the nine different types of nucleotide sugars used in human glycosylation. Dysfunction in NST activity is implicated in many diseases, yet little was known about the molecular details of how NSTs selectively recognize and transport their substrates. These structures reveal for the first time the principles of how mCST can selectively bind and transport CMP and CMP-coupled sugars versus other nucleotides and nucleotide-coupled sugars. These results provide a framework for understanding the effects of disease-causing mutations and the mechanisms of this diverse family of transporters.
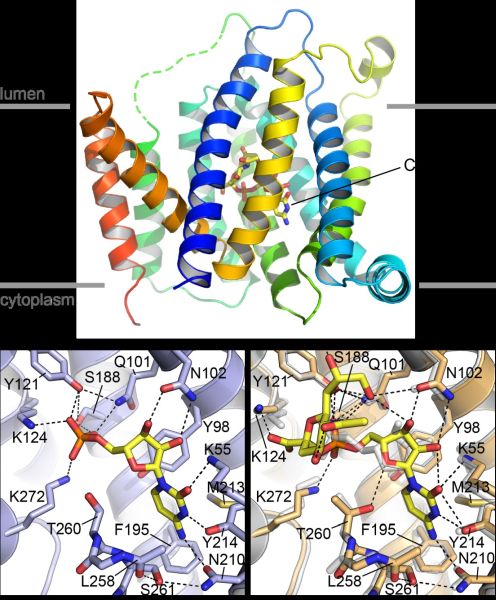 |
Figure: The ribbon representation on the top shows the structure of mCST in complex with CMP-Sia. The protein is colored in a rainbow pattern from blue (N-term) to red (C-term). The binding site for CMP-Sia is indicated. The approximate boundary of the Golgi membrane is also indicated. The bottom panels show the detailed substrate-protein interactions for CMP (left) and CMP-Sia (right). In the bottom right panel, the CMP-Sia-bound mCST structure is shown in light orange with the CMP-bound structure overlaid in light gray for comparison. |
Citation: Ahuja, S, Whorton, MR, Structural basis for mammalian nucleotide sugar transport, eLIFE J 8, e45221-1-e45221-27 (2019). DOI: 10.7554/eLife.45221